Imagine if fixing a broken bone or a damaged heart was as simple as replacing a part in a car. You wouldn’t need to wait for a donor or rely on metal plates; you would simply print a new, living part that fits perfectly. While this sounds like science fiction, 3D bioprinting is rapidly turning this concept into a medical reality.
Traditional medicine often relies on “inert” implants: metal or plastic pieces that hold the body together but don’t actually become part of it. Bioprinting takes a different approach. It uses living cells and biological materials to create structures that the body can adopt, heal, and eventually replace with its own tissue.
Here is how scientists are teaching printers to build living tissue, from hard bones to complex brain networks.
What Is 3D Bioprinting?
To understand bioprinting, think of a standard inkjet printer. Instead of ink cartridges filled with red, blue, and yellow dye, a bioprinter uses bioink.

Bioink is usually a mixture of two things:
1. Living Cells: These can be stem cells taken from fat or bone marrow.
2. A Scaffold Material: A gel or polymer that holds the cells in place, much like the frame of a house holds the bricks.
The printer lays this mixture down layer by layer. However, unlike printing a plastic toy, keeping these cells alive during and after the printing process is incredibly difficult. They need food (nutrients), air (oxygen), and a specific environment to survive.
The Challenge of Printing Bone
Bone seems simple, it is hard and structural, but it is actually a complex living tissue that constantly rebuilds itself. When printing bone, scientists face a contradiction: the material needs to be hard enough to support weight, but porous (full of holes) enough for cells to move in and grow.
The “Smart” Scaffold Solution
Researchers are now creating “smart” scaffolds that do more than just sit there. A 2024 review in Advanced Materials highlights scaffolds that can react to their environment, releasing antibacterial agents or growth factors when triggered.
One popular material for this is PLGA (polylactic-co-glycolic acid). It is biodegradable, meaning it slowly dissolves as the body builds new bone to replace it. A 2022 study detailed how PLGA can be mixed with ceramics to mimic natural bone strength while allowing the body to absorb the scaffold over time.
Using “Shape Memory” Materials (4D Printing)
This is where things get even more futuristic. 4D printing adds a fourth dimension: time. These printed objects can change their shape after they are printed, usually in response to heat or water.
Imagine a bone scaffold printed as a thin rod. A surgeon inserts it into a damaged area through a tiny hole. Once inside the body, the warmth causes the rod to expand and lock into the shape of the missing bone. A 2023 review explains how these shape memory materials could revolutionize minimally invasive surgeries, allowing large repairs through small incisions.
The “Blood Vessel” Hurdle
The biggest obstacle in tissue engineering is keeping the center of the tissue alive. In your body, no cell is more than a hair’s width away from a blood vessel. If you print a thick chunk of tissue without blood vessels, the cells in the middle starve and die. This is called necrosis.
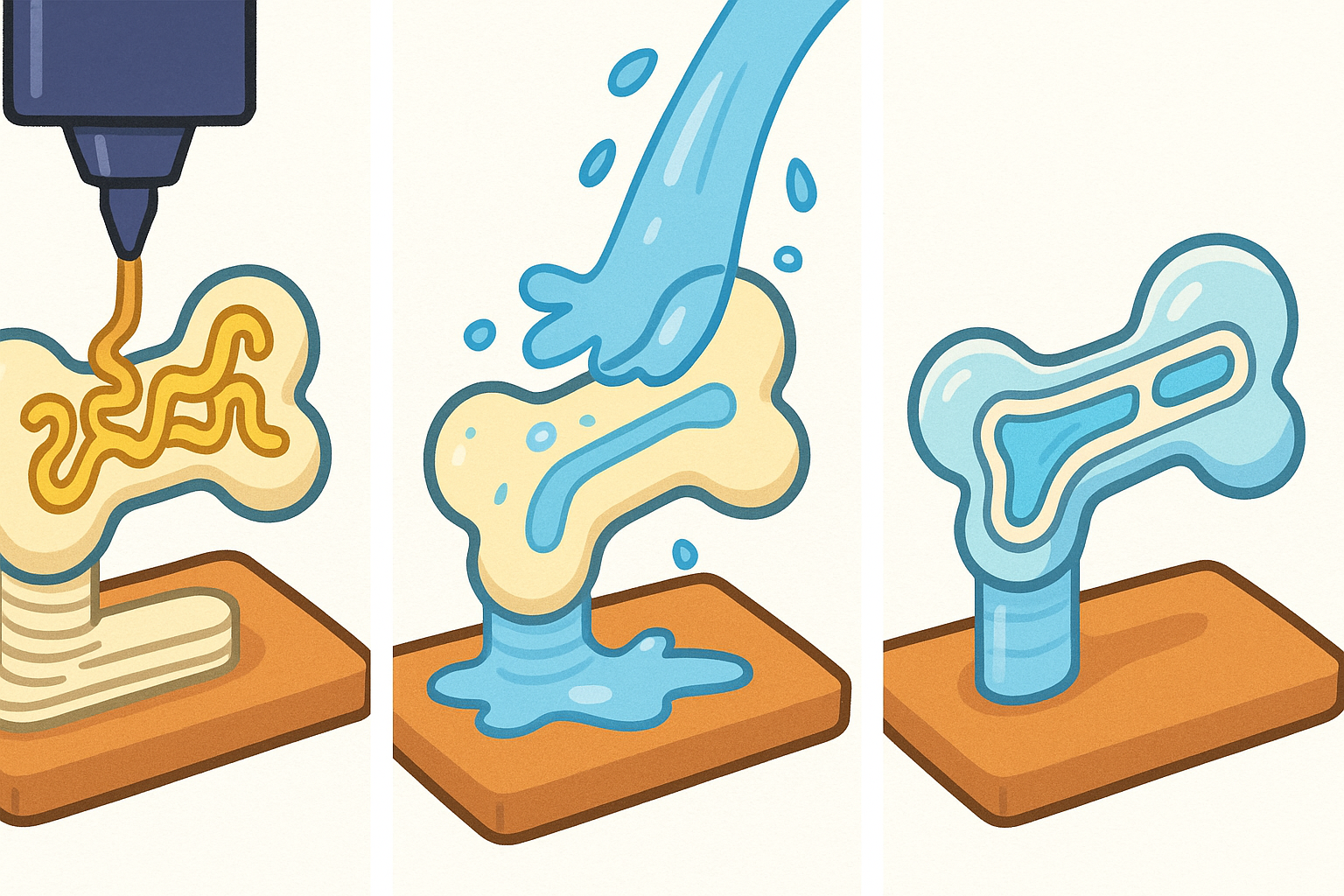
To solve this, scientists are using a technique called dual 3D printing.
In a 2021 study in Acta Biomaterialia, researchers printed a bone scaffold using a hard material, but they simultaneously printed a “sacrificial” ink inside it. This sacrificial ink acted like a placeholder. Once the structure was solid, they dissolved the sacrificial ink, leaving behind perfect, hollow channels. They then flushed these channels with endothelial cells (the cells that line blood vessels), effectively creating a working plumbing system inside the artificial bone.
Printing Brain and Nerve Tissue
Printing bone is one thing; printing the brain is entirely different. The brain relies on precise connections between neurons. If the cells are just thrown together randomly, they cannot communicate.
Recently, scientists achieved a breakthrough in printing functional neural tissue. A 2024 study in Cell Stem Cell described a method to print layers of neural progenitor cells (baby brain cells) using a special fibrin gel.
Instead of stacking layers vertically (like a cake), they printed bands horizontally. This allowed the neurons to grow extensions (axons) and physically connect with cells in neighboring bands. They successfully created a tissue where “cortical” neurons connected to “striatal” neurons, mimicking the actual wiring of the human brain. This could be a powerful tool for studying diseases like Parkinson’s or Alzheimer’s without using animal models.
Fixing the Heart
The heart presents a unique challenge: it moves. A printed heart patch must be elastic enough to stretch with every beat but strong enough not to tear. It also needs to conduct electricity so the cells beat in rhythm.
A 2021 review in Theranostics outlines how scientists are mixing conductive materials, like gold nanorods or carbon nanotubes, into the bioink. This ensures that the electrical signal from the heart travels through the patch, causing the printed cells to contract in sync with the patient’s own heart.
The Role of “Ghost” Tissues
Sometimes, the best material isn’t man-made at all. Scientists can take natural tissue (like fat or bone) and wash away all the cells using detergents. This leaves behind a white, sponge-like structure called the Decellularized Extracellular Matrix (dECM).
Think of dECM as a ghost of the original tissue. It retains the complex structure and proteins of the original organ but has no DNA to cause rejection.
- Fat Tissue: A 2015 study used dECM from fat tissue to print soft, dome-shaped constructs. When implanted in mice, these structures integrated with the host and spurred the growth of new blood vessels.
- Cartilage & Bone: A 2023 study utilized dECM from both cartilage and bone to print a dual-layered scaffold. This mimicked the natural transition from soft joint cartilage to hard bone, a complex interface that is notoriously difficult to repair.
The Bottom Line
3D bioprinting has moved beyond simple plastic models. By combining advanced polymers, shape-shifting 4D materials, and “ghost” tissues (dECM), researchers are creating implants that the body can actually inhabit and heal. While we cannot yet print a fully functional heart for transplant, we can print patches, bone grafts, and neural tissues that integrate with the body in ways metal and plastic never could.
Key Takeaways
- Bioinks combine living cells with a scaffold material to create printable tissue.
- Vascularization (creating blood vessels) is essential; without it, thick printed tissues die.
- 4D Printing allows implants to change shape after surgery, enabling minimally invasive procedures.
- dECM (natural tissue with cells removed) provides the most realistic environment for new cells to grow.
Quick Reference: Key Studies
| Application | Key Finding | Source |
|---|---|---|
| Brain | Printed neurons formed functional connections between cortical and striatal layers. | PMID 38306994 |
| Vascular Bone | Used a “sacrificial” ink to create hollow channels for blood vessels inside printed bone. | PMID 33454383 |
| 4D Printing | Shape-memory materials allow scaffolds to expand or change shape inside the body. | PMID 36614258 |
| Cartilage/Bone | Printed a bilayer scaffold using dECM and exosomes to repair joint defects. | PMID 37424038 |
| Soft Tissue | Decellularized fat tissue (dECM) was used as ink to print soft tissue implants. | PMID 26056727 |
| Heart | Conductive materials (gold/carbon) in bioink help printed heart patches beat in rhythm. | PMID 34335973 |
Last updated: March 2026
This article synthesizes findings from peer-reviewed research. It is for educational purposes only and does not constitute medical advice. Consult a healthcare provider before starting any new regimen.




Leave a Reply