Have you ever noticed that two people can eat the exact same extra portions over a holiday season, but only one seems to gain weight? Or perhaps you have wondered why some people carry extra weight in their hips, while others carry it in their bellies.
For a long time, weight gain was viewed as a simple math equation of calories in versus calories out. While it is true that a calorie surplus is required to gain weight, science shows that the human body is far more complex.
Research reveals that when we consistently overeat, our bodies do not respond uniformly. Instead, our genetics, our early childhood environment, and our cellular defense mechanisms completely alter how our bodies process, store, and react to those extra calories.
This article explores what decades of scientific research actually tells us about long-term overfeeding, starting with one of the most famous nutrition studies ever conducted.
How Do Genes Affect Weight Gain From Overeating?
To truly understand how the human body responds to overfeeding, scientists needed to control every variable. They needed to feed people the exact same amount of extra food, monitor their activity, and see what happened.
A landmark 1990 study in The New England Journal of Medicine did exactly this. Researchers gathered 12 pairs of healthy, identical young male twins. For 100 days, they lived in a closed dormitory where their diet and activity were strictly monitored.
The researchers fed every single man exactly 1,000 extra calories a day above what they needed to maintain their weight.
If weight gain were purely a matter of math, every man should have gained the exact same amount of weight. Instead, the results were astonishing:
- The amount of weight gained varied massively between different people, ranging from 9.5 pounds to 29 pounds.
- However, within each pair of identical twins, the weight gain was almost exactly the same.
- The twins also gained fat in the exact same areas of their bodies.

This proved that while a calorie surplus causes weight gain, your genes dictate how much weight you gain and where that weight goes.
Related: What Science Actually Says About Weight Loss and Obesity
The Genes That Control Fat Storage
Following the twin study, scientists spent years analyzing the DNA of the participants to figure out exactly which genes were responsible for these differences. A 2004 review in Obesity Reviews summarized these findings, showing that specific gene variations act like traffic cops, directing extra calories to different parts of the body.
Here is what the research uncovered about specific genes:
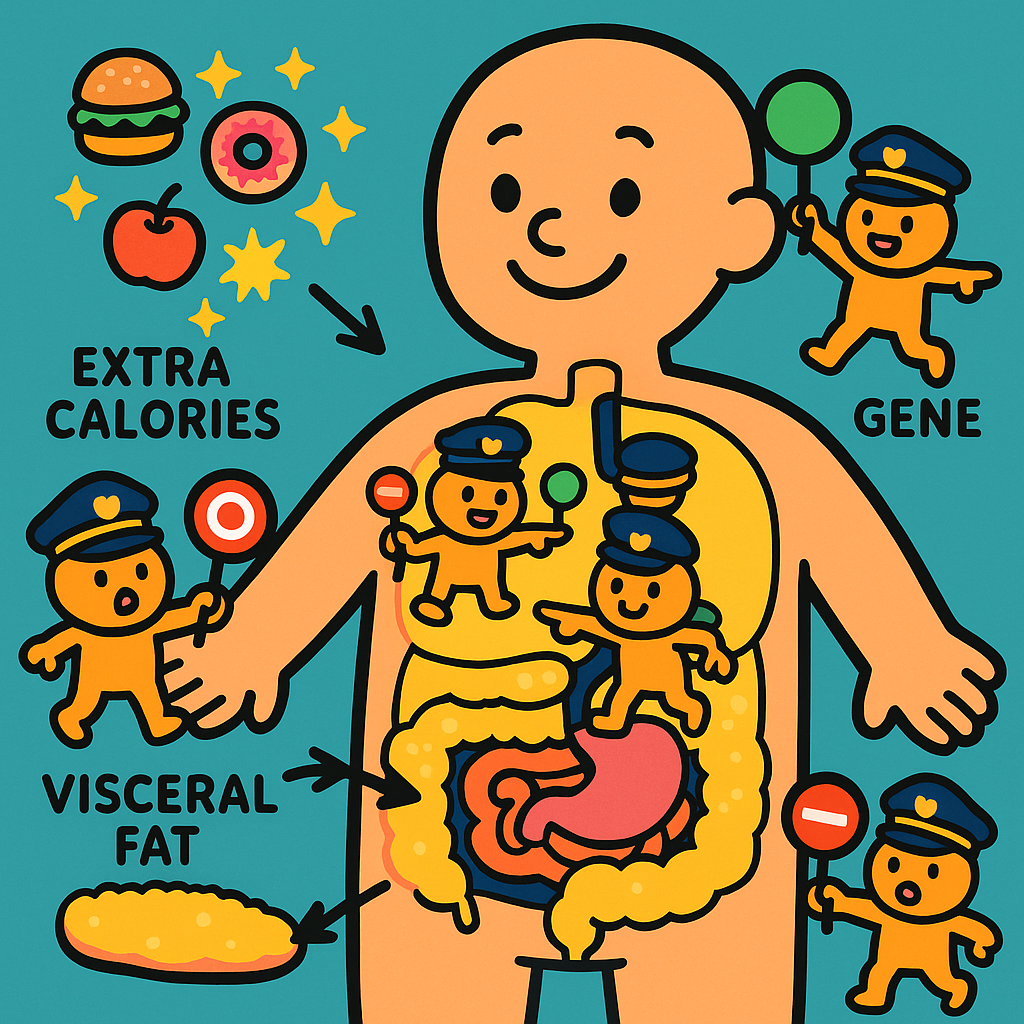
- The ADRB2 Gene: This gene influences how much fat you store just under your skin. A 2001 study in the International Journal of Obesity found that a specific variation of this gene caused certain men to gain significantly more weight and store much more fat directly under the skin compared to men without the variation.
- The Resistin and Adipsin Genes: These genes influence belly fat. A 2004 study in the European Journal of Clinical Nutrition found that a variation in the resistin gene caused individuals to store more extra calories as visceral fat (VISS-er-uhl fat). Visceral fat is the fat stored deep inside the belly around vital organs, and it is strongly linked to heart disease. Similarly, a 2003 study in the same journal found that variants in the adipsin gene also predicted higher gains in abdominal fat.
- The LPL Gene: Lipoprotein lipase (LPL) is an enzyme that helps clear fat from the bloodstream. A 2002 study in the Journal of Internal Medicine found that individuals with a specific LPL gene variant had a harder time clearing fats from their blood when overfed. This led to a drop in their “good” HDL cholesterol and an increase in blood triglycerides.
- The CETP Gene: This gene also plays a major role in cholesterol. A 2008 study in Atherosclerosis found that variations in this gene contributed to undesirable changes in cholesterol levels when exposed to excessive calorie consumption.
How Early Life Overfeeding Creates a Permanent Blueprint
While your genes provide the initial blueprint for how you handle food, your environment during infancy also plays a massive role.
Because scientists cannot ethically overfeed human infants to study long-term effects, they rely on rodent models. Researchers use a “small litter” method. By reducing the number of pups a mother rat nurses, the remaining pups get far more milk. This creates a state of early postnatal overfeeding.
A 2013 review in The Journal of Nutrition summarized the lifelong consequences of this early overfeeding. When infant rodents are overfed, it permanently wires their metabolism and brain in several ways:
1. Hormone Resistance
Overfeeding in infancy causes a rapid spike in leptin and insulin. Leptin is the hormone that tells the brain you are full. When the developing brain is flooded with leptin early on, it stops listening to the signal. This is called leptin resistance (LEP-tin ree-ZIS-tuhns). As a result, rodents overfed in infancy grow into adults who naturally eat more because their brains never properly receive the “I am full” signal.
2. Changes in the Brain’s Immune System
One of the most surprising discoveries is how early overfeeding affects the brain’s immune cells, known as microglia (my-kroh-GLEE-uh).
A 2020 study in Brain, behavior, and immunity found that early life overfeeding permanently “primes” these microglia, keeping them in a highly sensitive, pro-inflammatory state. A 2014 study in the same journal showed that this microglial activation happens specifically in the hypothalamus, the part of the brain that controls hunger and metabolism.
3. Exaggerated Responses to Illness
Because the immune system is permanently placed on high alert by early overfeeding, the body overreacts to future infections. A 2012 study in the Journal of Neuroendocrinology exposed adult rats (who had been overfed as infants) to a bacterial toxin. The overfed rats developed a significantly higher fever and released far more stress hormones than rats that were fed normally in infancy. A 2017 study in the same journal confirmed that this exaggerated immune response is directly tied to the primed microglia in the brain.
In short, overeating during the critical window of early development does not just add fat. It fundamentally alters the brain’s hunger signals and immune system for life.
How This Might Work: The Body’s Defense Mechanisms
When faced with a long-term surplus of calories, a healthy body does not just passively store fat. It actively tries to defend its normal weight by increasing energy expenditure.
One way it does this is through Brown Adipose Tissue (brown AD-ih-pohs TISH-oo), commonly called brown fat. Unlike white fat, which stores energy, brown fat burns energy to create heat in a process called thermogenesis (thur-moh-JEN-uh-sis).

A 2018 study in The Journal of Experimental Biology looked at what happens to brown fat when rats are overfed a sugary sucrose solution for three weeks. The researchers found that the rats did not gain as much weight as expected.
When they looked closer, they found that the brown fat had adapted. The cells had increased their number of mitochondria (the powerhouses of the cell) and boosted their levels of UCP1, a specific protein that allows cells to burn calories as heat. The body essentially tried to burn off the excess sugar by turning it into body heat to prevent excessive weight gain.
Related: What Science Actually Says About Blood Sugar After Meals
Who Needs Caution: Overfeeding in Critical Illness
While a healthy body has mechanisms like brown fat to handle extra calories, the rules completely change when a person is critically ill.
In intensive care units (ICUs), patients often lose muscle mass rapidly. Historically, doctors believed that providing high-calorie nutritional therapy would prevent this muscle loss. However, modern research shows that overfeeding critically ill patients is actually dangerous.
A 2024 review in the Journal of Intensive Care and a 2019 review in Critical Care explain that during a severe illness, the body enters an extreme state of stress. The body begins breaking down its own tissues for energy to fuel the immune system.
Providing excess calories through feeding tubes does not stop this internal breakdown. Instead, it overloads the patient’s system. The body cannot process the excess nutrients, which can lead to severe spikes in blood sugar, liver stress, and delayed recovery. In the ICU, doctors must carefully avoid energy overfeeding and slowly introduce nutrition only as the patient stabilizes.
Common Questions About Long-Term Overfeeding
Does overeating permanently stretch your stomach?
While the stomach can expand to accommodate large meals, it naturally shrinks back to its normal size once food is digested. Long-term overfeeding alters hormone signals and brain wiring regarding hunger, rather than permanently stretching the physical stomach organ.
Why do I feel hot after eating a massive meal?
This is known as diet-induced thermogenesis. As shown in the brown fat studies, your body ramps up heat production to help process the sudden influx of calories and nutrients, attempting to burn off a portion of the excess energy.
Can you fix leptin resistance caused by early childhood overfeeding?
Research in rodents shows that it is very difficult to completely reverse early-life metabolic programming. However, healthy dietary changes, regular physical activity, and weight management in adulthood can significantly improve how the body responds to insulin and leptin, even if the underlying genetic or childhood blueprint remains.
The Bottom Line
The science of long-term overfeeding shows us that weight gain is not a uniform process.
- Genetics matter: If you overeat, your genes determine how much of that energy becomes fat, whether it is stored safely under the skin or dangerously around your organs, and how your blood cholesterol reacts.
- Early life matters: Overfeeding during infancy can permanently alter the brain’s hunger signals and place the immune system in a chronic state of inflammation.
- The body fights back: Healthy bodies will attempt to burn off excess calories by generating heat through brown fat.
- Context matters: While healthy individuals can adapt to a calorie surplus, critically ill patients can be severely harmed by overfeeding.
Understanding these mechanisms helps remove the stigma around weight gain. It highlights that our bodies are highly complex systems reacting to our environment based on a blueprint written in our DNA and shaped during our earliest days of life.
Quick Reference: Key Studies
| Study Focus | Key Finding | Source |
|---|---|---|
| Twin Overfeeding Study | Identical twins gained the exact same amount of weight and fat distribution when overfed by 1000 calories/day, proving a strong genetic link. | PMID 2336074 |
| Gene Variants & Fat Storage | Variations in the Resistin, Adipsin, and ADRB2 genes dictate whether excess calories are stored as visceral or subcutaneous fat. | PMID 15042134, PMID 12947425, PMID 11753578 |
| Early Life Overfeeding | Overfeeding in infancy permanently alters brain wiring, causing leptin resistance and lifelong weight gain in rodents. | PMID 23446961 |
| Immune System Priming | Early life overfeeding places brain immune cells (microglia) in a permanent pro-inflammatory state, causing exaggerated fever responses to illness. | PMID 32088312, PMID 22175701 |
| Brown Fat Adaptation | Long-term sucrose overfeeding activates brown adipose tissue to burn excess calories as heat. | PMID 29496784 |
| ICU Overfeeding Risks | Overfeeding critically ill patients is dangerous because the stressed body cannot process excess calories. | PMID 39075627, PMID 31752979 |
Last updated: March 2026
This article synthesizes findings from peer-reviewed research. It is for educational purposes only and does not constitute medical advice. Consult a healthcare provider before starting any new regimen.



Leave a Reply