Your immune system is like a highly trained security force. Its primary job is to patrol your body, recognize foreign invaders like viruses or bacteria, and eliminate them. The key detectives in this security force are T-cells (TEE-sells), a type of white blood cell that hunts down abnormal cells.
However, cancer cells are masters of disguise. They can hide their identifying markers, allowing them to slip past T-cells unnoticed and multiply. For decades, researchers have looked for ways to help the immune system see through this disguise.
Enter Chimeric Antigen Receptor (ky-MARE-ik AN-tih-jen ree-SEP-tor) T-cell therapy, commonly known as CAR-T cell therapy. Often described by researchers as a “living drug,” this treatment involves extracting a patient’s own T-cells, genetically reprogramming them in a lab to recognize specific cancer disguises, and returning them to the body to hunt down the disease.
While this approach has shown remarkable success in treating certain blood cancers, it still faces significant hurdles when dealing with solid tumors like breast or lung cancer. Here is what the latest scientific research says about how CAR-T cell therapy works, where it succeeds, and the challenges that remain.
What is CAR-T Cell Therapy?
To understand CAR-T therapy, it helps to know how cells communicate. Cells have specific protein markers on their surfaces called antigens. T-cells use receptors to “read” these antigens and determine whether a cell is friendly or dangerous.
According to a 2024 review in Molecular Medicine Reports, CAR-T therapy essentially gives T-cells a new set of augmented-reality glasses. The process involves several distinct steps:
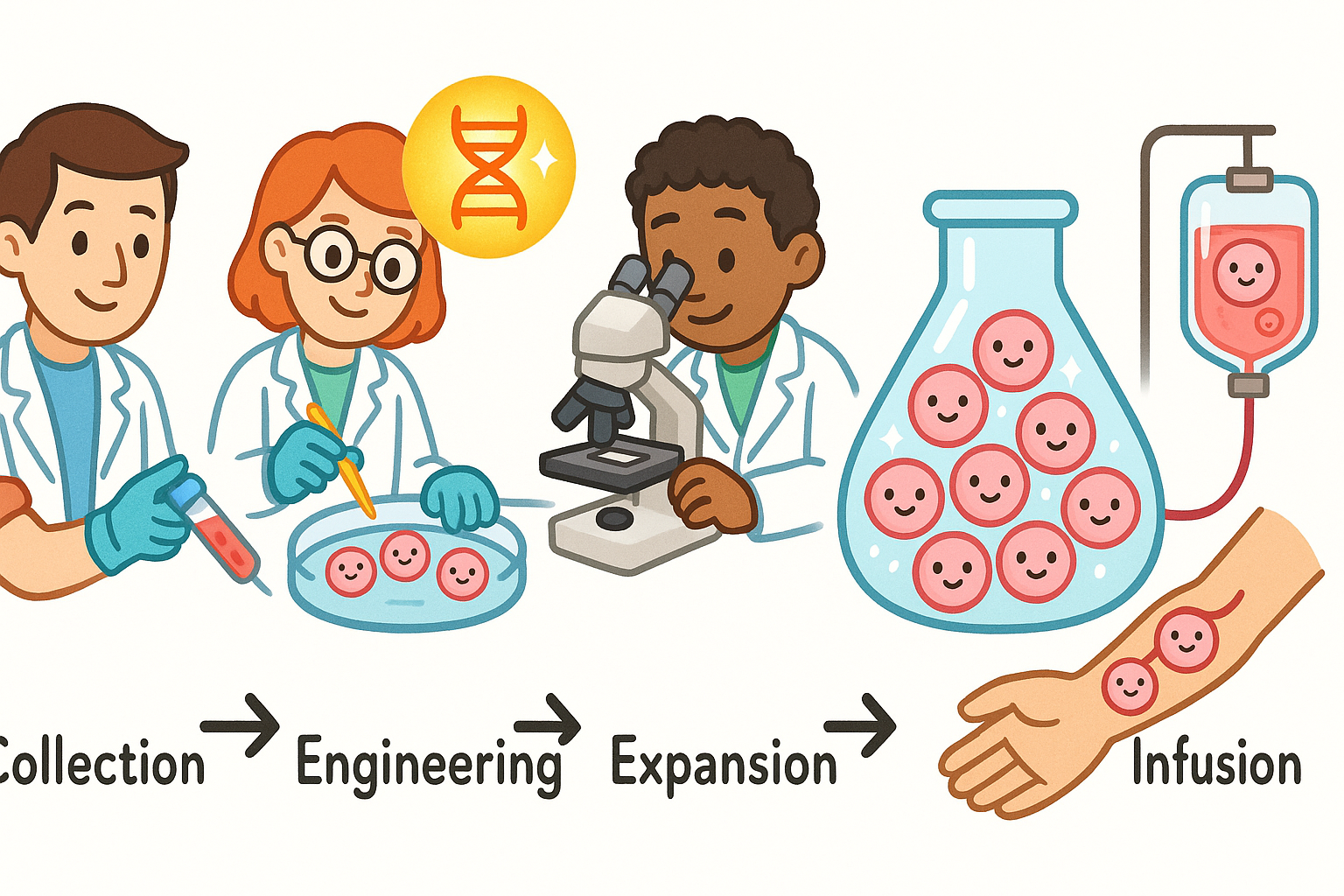

1. Collection: Doctors draw blood from the patient and separate out the T-cells.
2. Engineering: In a laboratory, scientists insert a new gene into the T-cells. This gene provides the instructions to build a Chimeric Antigen Receptor (CAR) on the cell’s surface.
3. Expansion: The newly engineered CAR-T cells are multiplied in the lab until there are millions of them.
4. Infusion: The patient receives a mild dose of chemotherapy to clear out old immune cells, making room for the new ones. The CAR-T cells are then infused back into the patient’s bloodstream.
Once inside the body, the CAR-T cells use their new receptors to latch onto specific antigens found on the surface of cancer cells. When they connect, the CAR-T cells release toxic chemicals that destroy the cancer cell.
Related: Does CRISPR Gene Therapy Actually Work? What the Science Says
Why It Works Well for Blood Cancers
CAR-T cell therapy has achieved its most notable successes in hematological (blood) cancers, such as leukemia, lymphoma, and multiple myeloma.
Blood cancer cells float freely in the bloodstream and lymphatic system, making them relatively easy for circulating CAR-T cells to find and attack. Furthermore, blood cancers often display very consistent antigens on their surfaces.
For example, a 2022 review in Current Problems in Cancer details how CAR-T therapies targeting an antigen called CD19 have transformed the treatment of B-cell lymphomas. In clinical trials for patients who had failed multiple prior treatments, CD19-targeted CAR-T cells achieved overall response rates between 52% and 82%, with many patients achieving complete remission.
Similarly, research has focused heavily on multiple myeloma, a cancer of plasma cells. A 2022 study in Pharmacology & Therapeutics highlights that targeting a specific marker called BCMA (B-cell maturation antigen) has yielded impressive results. Clinical trials for BCMA-targeted CAR-T therapies have shown response rates ranging from 73% to nearly 100% in heavily pretreated patients.
However, researchers note that while initial response rates are high, the durability of the response can vary. Some patients experience a relapse if their cancer cells mutate to stop expressing the target antigen, a process known as “antigen escape.”
The Challenge with Solid Tumors
While CAR-T therapy has proven highly effective for blood cancers, translating that success to solid tumors (like breast, lung, or brain cancer) has been difficult.
A 2024 paper in Frontiers in Immunology outlines several reasons why solid tumors are much harder to treat:
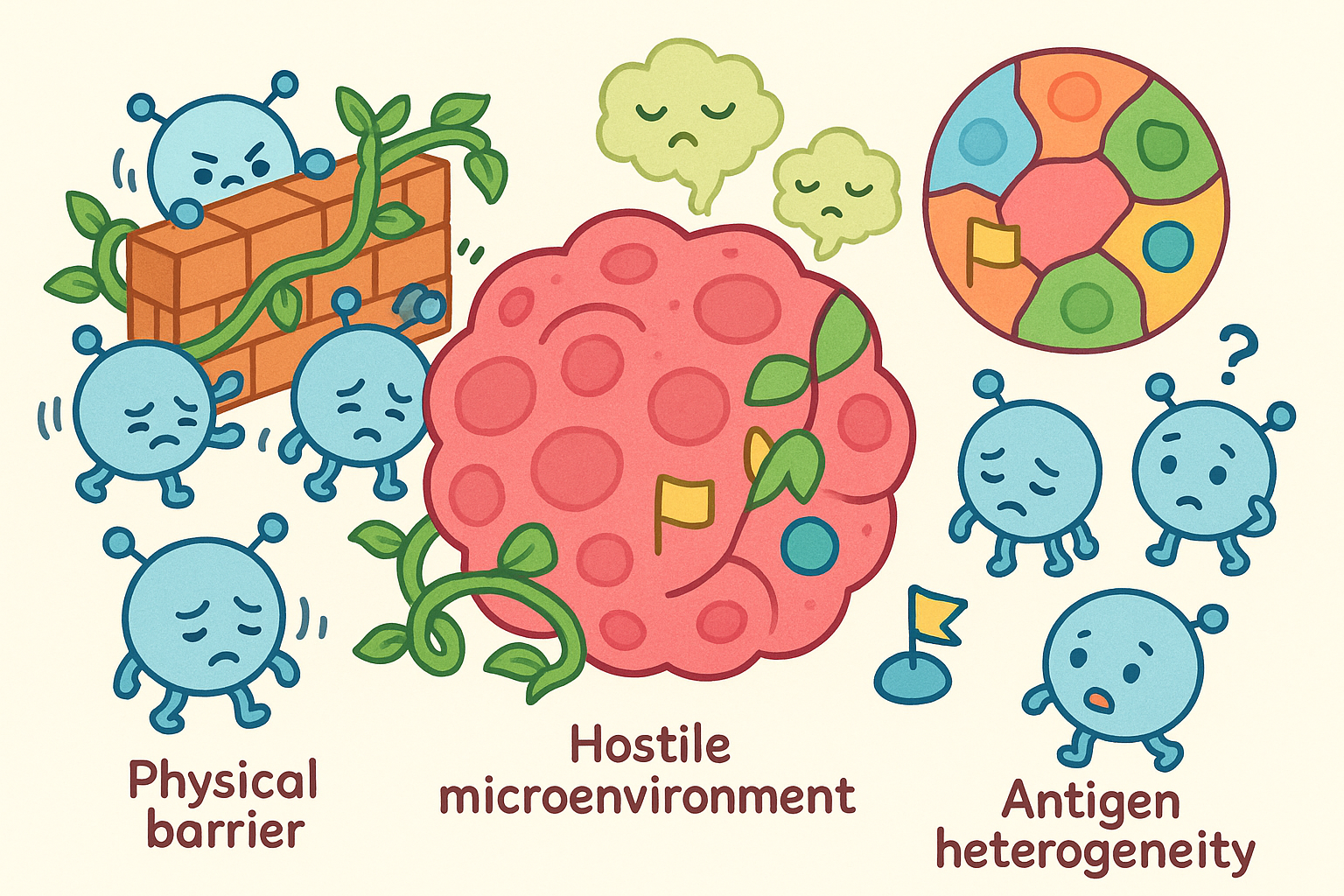
- The Physical Barrier: Solid tumors build dense physical structures around themselves. CAR-T cells struggle to penetrate this dense tissue to reach the cancer cells inside.
- The Tumor Microenvironment: Solid tumors create a hostile chemical environment around themselves. They release signals that suppress the immune system, effectively putting CAR-T cells to sleep or causing them to become exhausted.
- Antigen Heterogeneity: Unlike blood cancers, which often display the same antigen on every cell, solid tumors are mixed. One part of a lung tumor might display a specific antigen, while another part of the exact same tumor does not. If a CAR-T cell is programmed to attack Antigen A, the cancer cells lacking Antigen A will survive and continue to grow.
Breast Cancer and Lung Cancer Trials
Researchers are actively searching for reliable antigens to target in solid tumors.
In breast cancer, a 2022 review in the International Journal of Biological Sciences notes that scientists are testing CAR-T cells designed to target HER2 and EGFR, which are proteins often overexpressed in certain breast cancer subtypes. Early laboratory and animal studies show that these targeted cells can shrink tumors, but clinical trials in humans are still in the very early stages.
In lung cancer, similar efforts are underway. A 2022 study in Thoracic Cancer highlights clinical trials targeting antigens like MSLN (mesothelin) and MUC1. However, progress is slow because these antigens are sometimes found in small amounts on healthy tissue. If CAR-T cells attack healthy lung tissue alongside the cancer, it can cause severe, life-threatening inflammation.
Who Needs Caution: Managing Side Effects
Because CAR-T therapy involves supercharging the immune system, it comes with significant risks. It is generally reserved for patients whose cancer has not responded to standard treatments like chemotherapy or radiation.
According to a 2021 review in Pathology, there are two primary side effects that require close medical monitoring:
1. Cytokine Release Syndrome (CRS): When CAR-T cells attack cancer, they release inflammatory proteins called cytokines. If too many cytokines are released at once, the body experiences a massive inflammatory response. Symptoms range from high fevers and chills to dangerous drops in blood pressure and organ failure.
2. Neurotoxicity (ICANS): Some patients experience confusion, difficulty speaking, tremors, or even seizures. The exact reason why CAR-T therapy affects the brain is still being studied, but it is believed to be related to the systemic inflammation caused by CRS.
Doctors have developed strict protocols to manage these side effects, often using specific medications to calm the immune system if it becomes overactive.
How Science is Improving the Therapy
To overcome the limitations of CAR-T therapy, especially in solid tumors, scientists are developing creative new strategies.
Combining CAR-T with Oncolytic Viruses
One approach involves using oncolytic viruses (on-co-LIT-ik), which are viruses genetically modified to infect and kill only cancer cells.
According to research in Frontiers in Immunology, injecting an oncolytic virus into a solid tumor can break open the tumor cells and release inflammatory signals. This effectively turns a “cold” tumor that the immune system ignores into a “hot” tumor that attracts immune cells. When combined with CAR-T therapy, the virus acts like a battering ram, breaking down the tumor’s physical and chemical defenses so the CAR-T cells can enter and finish the job.
Armored CARs and Regional Delivery
To combat the hostile tumor microenvironment, researchers are designing “armored” CAR-T cells. These next-generation cells are engineered not just to recognize cancer, but also to secrete their own immune-boosting chemicals to keep themselves active in harsh conditions.
Another strategy is changing how the cells are delivered. Instead of injecting CAR-T cells into the bloodstream and hoping they find their way to a solid tumor, doctors are trying regional delivery. A 2021 study in Lung Cancer focusing on malignant pleural mesothelioma (a cancer of the lung lining) found that injecting CAR-T cells directly into the chest cavity (rather than the bloodstream) resulted in much better tumor penetration and longer-lasting cancer suppression in animal models.
Related: How MRI Is Changing the Way Doctors Find Cancer
Common Questions About CAR-T Cell Therapy
Is CAR-T therapy the same as chemotherapy?
No. Chemotherapy uses toxic chemicals to kill rapidly dividing cells throughout the body. CAR-T therapy uses your own genetically modified immune cells to specifically target and destroy cancer cells.
How long does it take to make CAR-T cells?
Currently, the manufacturing process takes about 3 to 4 weeks. Blood is drawn, sent to a specialized laboratory for genetic modification and multiplication, and then shipped back to the hospital for infusion.
Can CAR-T therapy cure cancer?
For some patients with specific types of blood cancer, CAR-T therapy has resulted in long-term, durable remissions that doctors cautiously refer to as cures. However, it does not work for everyone, and researchers are still working to improve its effectiveness for solid tumors.
The Bottom Line
CAR-T cell therapy represents a significant shift in how we approach cancer treatment. By rewriting the genetic code of a patient’s own immune cells, science has found a way to target specific blood cancers with remarkable accuracy.
However, the evidence is clear that we are still in the early chapters of this research. While therapies for leukemia, lymphoma, and multiple myeloma are currently available and saving lives, the application of CAR-T for solid tumors remains largely experimental. Overcoming the physical barriers of solid tumors, preventing antigen escape, and managing severe immune side effects are the primary hurdles scientists are working to solve today.
Quick Reference: Key Studies
| Study Focus | Key Finding | Source |
|---|---|---|
| General Overview | CAR-T modifies patient T-cells to target cancer, showing high success in blood cancers but facing hurdles in solid tumors. | PMID 38275119 |
| Multiple Myeloma | BCMA-targeted CAR-T therapies show response rates up to 100% in heavily pretreated patients, though relapse remains a challenge. | PMID 34582835 |
| B-Cell Lymphoma | CD19-targeted CAR-T therapies provide durable remissions for patients who failed prior chemotherapy. | PMID 35012754 |
| Solid Tumors & Viruses | Combining CAR-T cells with cancer-killing (oncolytic) viruses helps break down the defenses of solid tumors. | PMID 39539554 |
| Lung Cancer | CAR-T targeting antigens like MSLN and EGFR shows preclinical promise but faces challenges with off-target toxicity on healthy tissue. | PMID 35289077 |
| Mesothelioma | Injecting CAR-T cells directly into the chest cavity (regional delivery) improves tumor penetration compared to intravenous delivery. | PMID 33972125 |
Last updated: March 2026
This article synthesizes findings from peer-reviewed research. It is for educational purposes only and does not constitute medical advice. Consult a healthcare provider before starting any new regimen.



Leave a Reply