Understanding the Genetic Instruction Manual
Our DNA acts like a massive instruction manual for the body. It contains billions of letters that tell our cells how to build muscles, process food, and fight off infections. Sometimes, a person is born with a single incorrect letter in this manual. This tiny typo can cause serious health conditions, such as sickle cell disease or muscular dystrophy.

For decades, medicine could mostly treat the symptoms of these conditions. Fixing the root cause seemed impossible. Then, scientists discovered a natural defense system inside bacteria that could be reprogrammed to edit human DNA. This system is called CRISPR.
While CRISPR has transformed laboratory research, moving it from a petri dish into a living human body involves complex biology. The challenge is no longer just how to cut the DNA, but how to safely deliver the microscopic tools to the exact right cells without causing unwanted damage.
How the Genetic Scissors Might Work
To understand how this technology works, it helps to learn a few key terms:
- CRISPR (KRIS-per) stands for Clustered Regularly Interspaced Short Palindromic Repeats. In nature, it is a memory system bacteria use to remember and fight off attacking viruses.
- Cas9 (KASS-nine) is a specific protein that acts like a pair of molecular scissors.
- Endonuclease (en-doh-NOO-klee-ace) is the scientific name for an enzyme that can cut through DNA strands.
The CRISPR-Cas9 system works in two parts. First, a piece of “guide RNA” acts like a GPS navigation system. It scans the billions of letters in the human genome to find an exact match. Second, once the guide RNA finds the target, the Cas9 protein cuts the DNA at that exact spot.

A 2023 paper in Frontiers of Medicine explains that this dual-RNA structure allows scientists to program the Cas9 scissors to target almost any DNA sequence. Once the DNA is cut, the cell naturally tries to repair the break. Scientists can use this repair process to either disable a harmful gene or paste in a corrected version of the gene.
What the Research Shows About CRISPR in Medicine
Clinical trials testing CRISPR in humans are already underway, and the results vary depending on the disease being treated.
Treating Blood Disorders
The most notable success for CRISPR so far involves blood disorders like sickle cell disease and beta-thalassemia. In these conditions, a genetic error causes the body to produce abnormal red blood cells.
A 2024 review in Cells highlights that the FDA recently approved the first CRISPR-based drug for these diseases, known as Casgevy. The treatment involves removing blood stem cells from the patient’s bone marrow, editing them in a laboratory to produce healthy hemoglobin, and then returning the cells to the patient. Early results show that this can drastically reduce the severe pain crises associated with sickle cell disease and reduce the need for blood transfusions.
Upgrading the Immune System to Fight Cancer
Cancer cells are notoriously good at hiding from the body’s immune system. They often press a “brake pedal” on immune cells, telling them to stop attacking.
Researchers are using CRISPR to remove these brakes. A 2019 review in Cancer Letters details how scientists can extract a patient’s T-cells (a type of white blood cell) and use CRISPR to knock out a gene called PD-1. Without the PD-1 brake, the T-cells become much more aggressive at hunting down and destroying tumor cells.
This approach is similar to other highly precise medical tools that allow doctors to target disease without harming surrounding healthy tissue. Related: How MRI Is Changing the Way Doctors Find Cancer
Addressing Inherited Eye Diseases
Treating diseases inside the body is much harder than editing blood cells in a lab. However, the human eye is a small, enclosed space, making it an ideal testing ground for gene therapy.
A 2022 study in the Journal of Nanobiotechnology outlines ongoing clinical trials using CRISPR to treat Leber congenital amaurosis (LCA10), a genetic disorder that causes childhood blindness. In this trial, the CRISPR tools are injected directly into the retina to cut out the genetic mutation causing the blindness.
Who Benefits Most Right Now
The current research clearly divides patients into two categories based on how the therapy is delivered.
Patients with blood disorders or certain immune-based cancers benefit the most from current CRISPR technology. This is because their treatments can be performed ex vivo (eks VEE-voh), meaning the cells are edited outside the body. Doctors can check the cells in the lab to ensure the edit was successful before returning them to the patient.
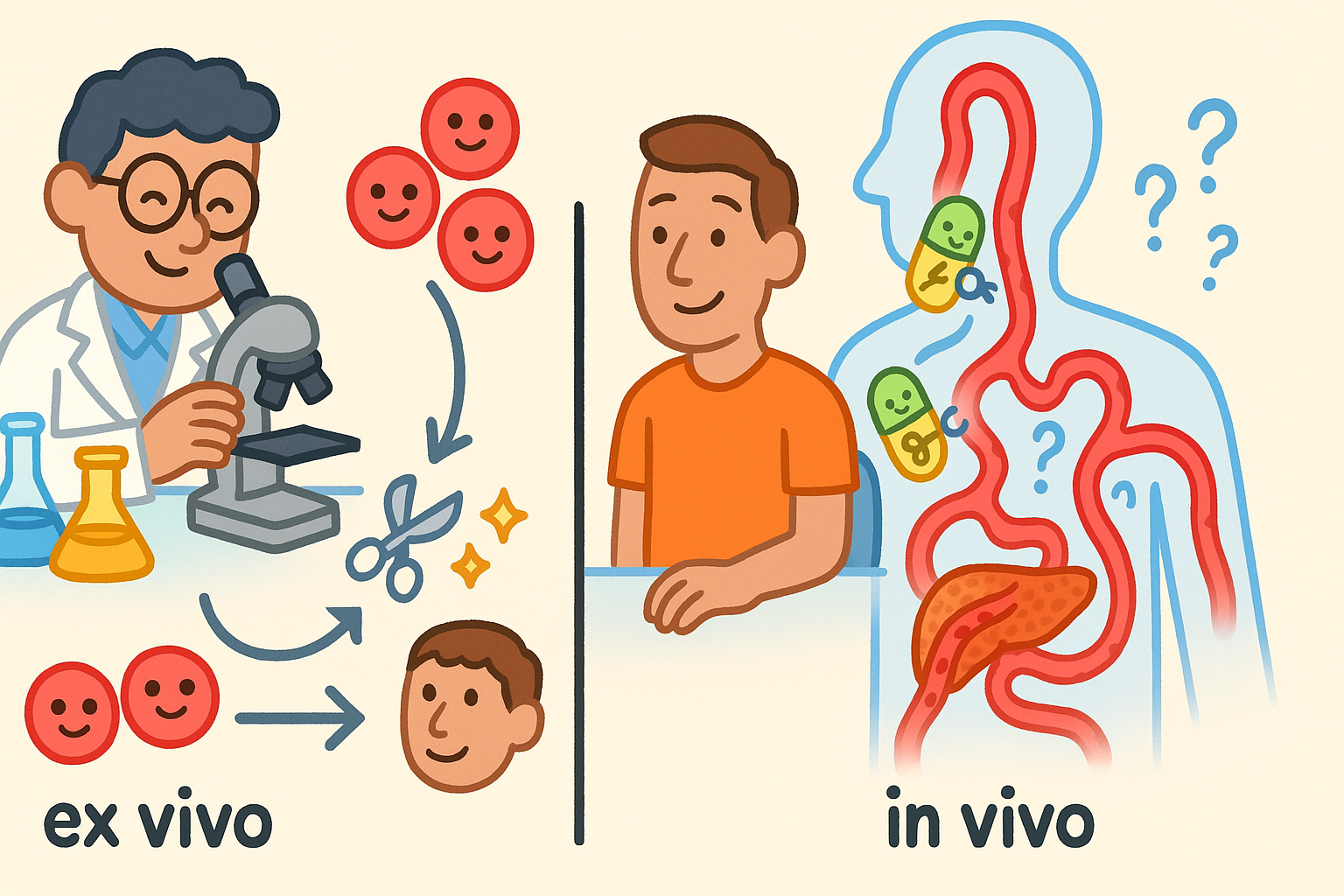
Patients with diseases affecting solid organs like the liver, heart, or muscles face a harder path. Their treatments must be performed in vivo (in VEE-voh), meaning the CRISPR tools must be injected into the bloodstream and find their way to the correct organ. This remains a major hurdle in the science today.
Where The Science Is Still Uncertain
While the concept of CRISPR is straightforward, the biological reality is messy. Researchers are actively working to solve several significant problems.
The Delivery Problem
If CRISPR is a package, the body needs a reliable postal service to deliver it. Right now, scientists often use hollowed-out viruses to carry the CRISPR instructions into cells. Adeno-associated viruses (AAV) are the most common choice.
However, a 2024 paper in AAPS PharmSciTech explains that viruses have limits. The Cas9 protein is physically large, making it difficult to stuff inside a small virus. Furthermore, the human immune system often recognizes these viruses as invaders and attacks them before they can deliver the gene-editing tools.
To solve this, scientists are testing non-viral delivery methods. A 2021 study in the International Journal of Molecular Sciences explores the use of extracellular vesicles. These are tiny, natural fat bubbles that cells use to communicate with each other. By packing CRISPR inside these natural bubbles, scientists hope to sneak the tools past the immune system.
Off-Target Effects
One of the biggest safety concerns is that Cas9 might cut the wrong piece of DNA. This is known as an off-target effect.
A 2020 review in Frontiers in Oncology notes that if CRISPR cuts a healthy gene, it could accidentally trigger cell damage or even lead to cancer. Scientists are constantly tweaking the guide RNA and engineering new versions of Cas9 to make the system more accurate.
The Stress of Broken DNA
Standard CRISPR works by completely severing both sides of the DNA ladder. This creates a double-strand break. While this is necessary for traditional gene editing, it can cause the cell to panic and respond unpredictably.
To avoid this, scientists have developed newer versions of the tool called base editors and prime editors. A 2023 study in Signal Transduction and Targeted Therapy describes base editing as a chemical eraser. Instead of cutting the whole DNA ladder, a base editor chemically changes a single letter (like turning a C into a T). This is much gentler on the cell and reduces the risk of unwanted mutations.
Common Questions About CRISPR
Is CRISPR being used to create designer babies?
No. Currently, all approved CRISPR therapies and mainstream clinical trials involve somatic cell editing. This means the genetic changes only affect the individual patient and cannot be passed down to their children. A 2018 paper in Current Issues in Molecular Biology emphasizes that editing human embryos (germline editing) remains highly controversial, ethically fraught, and illegal in many countries.
Is a CRISPR treatment a permanent cure?
For many genetic diseases, the goal is a permanent cure. Because CRISPR alters the actual DNA blueprint of the cell, those changes are permanent. If the edited cells survive and multiply, the patient may never need treatment for that specific genetic error again. However, long-term studies are still tracking patients to see how long the benefits last.
The Bottom Line
CRISPR has fundamentally changed how scientists approach genetic diseases. We now have the ability to locate and edit specific typos in the human genome. The evidence shows that this technology is highly effective for diseases where cells can be removed, edited in a lab, and returned to the body, such as sickle cell disease.
However, significant uncertainties remain regarding how to safely deliver these tools to internal organs without triggering the immune system or accidentally cutting the wrong DNA. As researchers develop smaller, more precise editing tools and better delivery vehicles, the list of treatable conditions will likely grow.
Quick Reference: Key Studies
| Study Focus | Key Finding | Source |
|---|---|---|
| Blood Disorders | Ex vivo CRISPR therapies (like Casgevy) successfully treat sickle cell disease and beta-thalassemia by restoring healthy hemoglobin. | PMID 38786024 |
| Cancer Immunotherapy | Knocking out the PD-1 gene in T-cells removes immune “brakes” and helps cells attack tumors more effectively. | PMID 30684591 |
| Viral Delivery Limits | AAV viral vectors are effective but limited by size constraints and potential immune system attacks. | PMID 39702810 |
| Base Editing | Newer CRISPR tools can change single DNA letters without completely breaking the DNA strand, reducing cellular stress. | PMID 36646687 |
| Off-Target Effects | Unintended DNA cuts remain a major hurdle, requiring highly engineered Cas9 variants to improve safety. | PMID 32850447 |
Last updated: March 2026
This article synthesizes findings from peer-reviewed research. It is for educational purposes only and does not constitute medical advice. Consult a healthcare provider before starting any new regimen.




Leave a Reply