Most of us think of brushing and flossing simply as ways to prevent cavities and keep our breath fresh. However, your mouth is actually a bustling ecosystem. It serves as the primary gateway to your body, hosting a massive community of microscopic life that interacts with your immune system, your digestive tract, and even your brain.

This community is known as the Oral microbiome (OR-al my-kro-BY-ohm), which includes all the bacteria, fungi, and viruses living in your mouth. While we often view bacteria as enemies to be washed away, a healthy oral microbiome is essential for your overall well-being.
Recent scientific research reveals that when the balance of bacteria in your mouth is disrupted, it can trigger a domino effect throughout your entire body. This article explores what science actually says about the bacteria in your mouth, how they travel to other organs, and what this means for your long-term health.
What Lives in a Healthy Mouth?
The human mouth is the second most diverse microbial environment in the body, right behind the gut. According to research published in the Journal of Microbiology & Biology Education, a healthy oral cavity is dominated by specific families of bacteria, such as Streptococcus, Veillonella, and Prevotella.
In a balanced state, these microbes are harmless and even helpful. They help break down food, protect against outside invaders, and maintain the health of your gums. For example, a 2026 preprint study looking at harmless species of Neisseria bacteria in the mouth found that these microbes are universally carried by healthy adults. Interestingly, the study noted that these harmless bacteria can sometimes act as an “early warning system” for scientists, as they can carry and share genes that help us understand how bacteria adapt to their environments.
However, the mouth is a unique environment. Unlike your skin or your intestinal lining, which constantly shed dead cells to remove attached bacteria, your teeth are hard, non-shedding surfaces. This allows bacteria to build up and form a Biofilm (BY-oh-film), which is a sticky, protective layer of microbes commonly known as plaque.
How Things Go Wrong: The “Leaky Gum” Concept
If you have ever heard of “leaky gut,” you might be familiar with the idea that a damaged intestinal lining can allow bacteria to enter the bloodstream. Scientists are now applying a similar concept to the mouth, often referred to as “leaky gum.”
A 2022 review in Cells outlines exactly how this happens. The anatomy of your gums creates a natural vulnerability. The small space between your tooth and your gum tissue is called the Gingival sulcus (JIN-jih-vul SUL-kus). At the bottom of this space is the Junctional epithelium (JUNK-shun-al ep-ih-THEE-lee-um), a delicate layer of tissue that attaches the gum to the tooth.
Because teeth do not shed their surface layer, biofilms can accumulate in the gingival sulcus over a lifetime if they are not physically removed by brushing and professional cleanings. When this bacterial layer grows too thick, it triggers an immune response. This leads to Periodontitis (per-ee-oh-don-TIE-tis), a severe gum infection that causes inflammation and tissue damage.
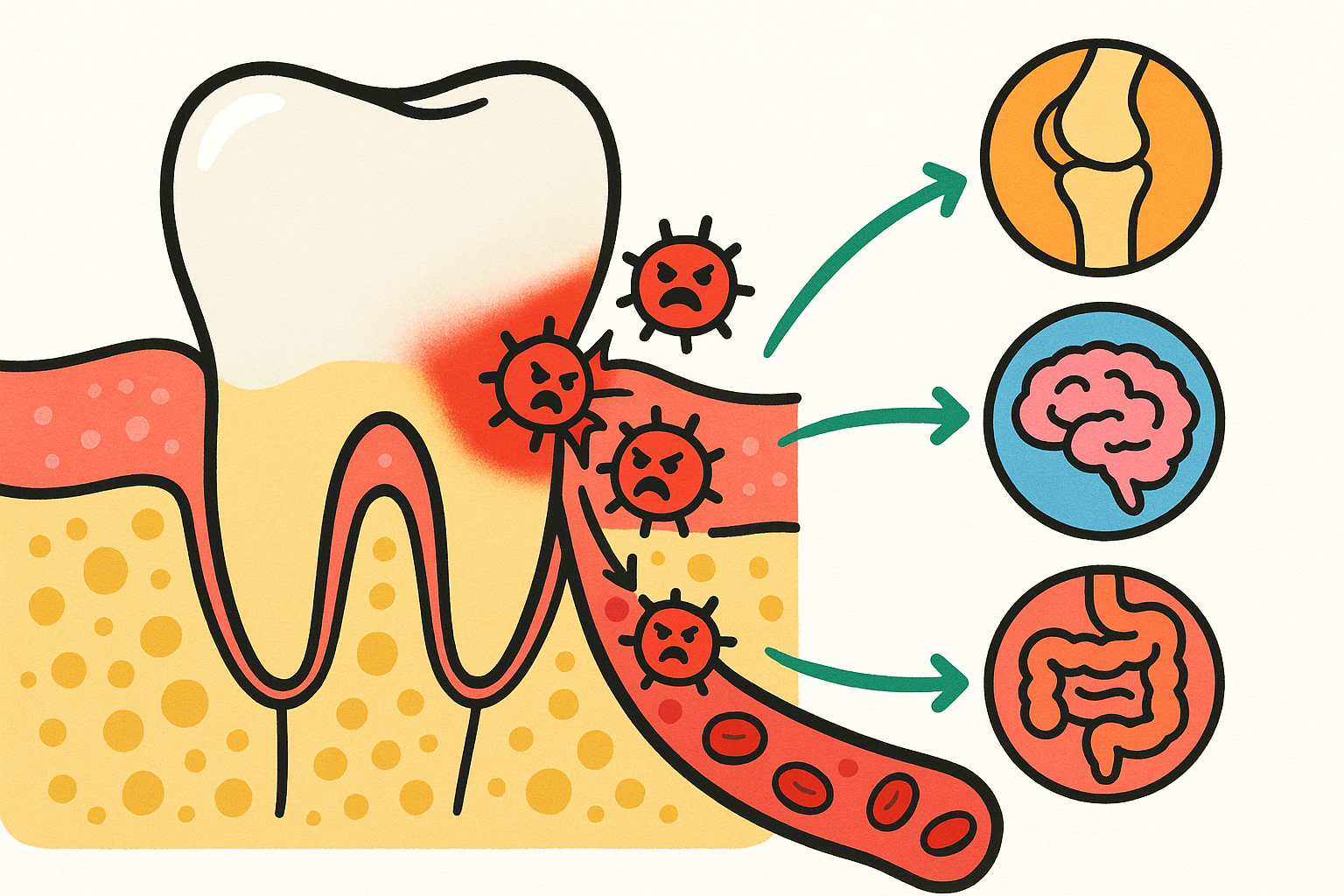
Once the junctional epithelium is damaged, the barrier is broken. The “leaky gum” allows oral bacteria and their inflammatory byproducts to slip directly into your bloodstream. From there, they can travel to almost any organ in your body.
The Oral-Gut Connection
The bloodstream is not the only way oral bacteria travel. Every time you swallow, you send thousands of oral bacteria down into your digestive tract.
Historically, scientists believed that stomach acid destroyed most of these bacteria before they could reach the intestines. However, a 2019 review in FEMS Microbiology Reviews highlights that certain resilient oral bacteria can survive the journey. Once they reach the intestines, they can disrupt the balance of your gut microbiome.
When oral bacteria colonize the gut, they can trigger chronic inflammatory reactions. This creates a direct line of communication between the health of your mouth and the health of your digestive system, often referred to as the oral-gut axis.
Related: Does Leaky Gut Syndrome Actually Exist? What the Latest Science Says
What the Research Shows: Systemic Diseases
When harmful oral bacteria escape the mouth through a “leaky gum” or by surviving the journey to the gut, they can contribute to several systemic health conditions.
Rheumatoid Arthritis
Rheumatoid arthritis is an autoimmune condition where the body mistakenly attacks its own joints. Research has found a surprisingly strong link between this joint disease and gum disease.
The 2019 review in FEMS Microbiology Reviews explains that a specific oral pathogen called Porphyromonas gingivalis (P. gingivalis) produces an enzyme that modifies human proteins. This modification process makes the proteins look “foreign” to the immune system. In genetically susceptible people, the immune system begins attacking these modified proteins, which can trigger or worsen the severe joint inflammation seen in rheumatoid arthritis.
Diabetes and Blood Sugar
The relationship between your oral microbiome and metabolic health is a two-way street. Research in the International Heart Journal explains that diabetes increases the severity and progression of gum disease. Conversely, severe gum disease increases insulin resistance and makes it harder for patients to control their blood sugar.
When oral bacteria enter the bloodstream, they promote a state of systemic inflammation. This constant, low-grade inflammation interferes with how your cells respond to insulin. The researchers noted that patients with poorly controlled diabetes had significantly higher rates of the harmful P. gingivalis bacteria in their mouths compared to those with well-controlled blood sugar.
Related: How Prediabetes Affects Your Body and Type 2 Diabetes Risk
Alzheimer’s Disease and Brain Health
Perhaps the most surprising area of oral microbiome research involves the brain. A 2020 study in Brain Sciences outlines how the gut-brain axis and oral bacteria might influence neurological diseases.
When “leaky gum” allows bacteria into the bloodstream, some microbes can cross the blood-brain barrier. Studies have found the DNA of oral bacteria, specifically P. gingivalis, inside the brains of Alzheimer’s patients. Once in the brain, these bacteria can activate specialized immune cells called microglia. While these cells normally protect the brain, chronic activation by oral bacteria can lead to neuroinflammation, which is a driving factor in the progression of Alzheimer’s disease.
Where the Science Is Still Uncertain
While the connections between the oral microbiome and systemic diseases are well-documented, scientists are still working to understand the exact chain of events.
The core tension in this field of research is determining correlation versus causation. For example, do oral bacteria directly cause Alzheimer’s disease, or do people in the early stages of cognitive decline simply struggle to maintain their dental hygiene, leading to an overgrowth of bad bacteria?
Animal studies strongly suggest that introducing oral pathogens directly causes systemic inflammation and disease progression. However, in humans, diseases like rheumatoid arthritis and Alzheimer’s develop over decades and involve genetics, diet, and lifestyle. Therefore, poor oral health is currently viewed as a significant contributing risk factor rather than the sole cause of these complex diseases.
Practical Guidance
Because the teeth provide a hard surface where biofilms can grow indefinitely, physical removal is the only way to manage the oral microbiome effectively.
- Disrupt the Biofilm: Regular brushing and flossing physically break up the bacterial colonies in the gingival sulcus before they can damage the tissue barrier.
- Manage Blood Sugar: Because high blood sugar impairs your body’s ability to heal and fight infection, maintaining metabolic health directly supports your gum tissue’s ability to keep bacteria out of your bloodstream.
- Professional Cleanings: Once a biofilm hardens into tartar, it cannot be removed at home. Professional cleanings are required to reset the bacterial balance.
The Bottom Line
Your oral microbiome is a complex community of microbes that plays a vital role in your overall health. While most of these bacteria are harmless, poor oral hygiene allows them to form thick biofilms that damage the delicate tissue barrier of your gums.
This “leaky gum” scenario allows bacteria to enter the bloodstream and the gut, sparking systemic inflammation. Current research clearly links this wandering oral bacteria to worsened outcomes in diabetes, rheumatoid arthritis, and neurodegenerative conditions. While we cannot yet say that gum disease is the sole cause of these conditions, keeping your oral microbiome balanced is a scientifically backed way to protect your body’s major organs.
Quick Reference: Key Studies
| Study Focus | Key Finding | Source |
|---|---|---|
| Leaky Gum and Systemic Disease | Oral bacteria can enter the bloodstream through damaged gum tissue, contributing to cardiovascular and neurodegenerative diseases. | PMID 35406643 |
| Rheumatoid Arthritis | Oral pathogens like P. gingivalis can modify proteins, triggering immune responses linked to rheumatoid arthritis. | PMID 30219863 |
| Diabetes and Periodontitis | There is a bidirectional relationship where diabetes worsens gum disease, and gum disease increases insulin resistance. | PMID 30068836 |
| Brain Health and Microbiome | Oral and gut bacteria can drive neuroinflammation, which is a key feature in the progression of Alzheimer’s disease. | PMID 33153085 |
| Healthy Oral Microbiome | A healthy mouth relies on a diverse balance of bacteria, which can be identified through DNA sequencing. | PMID 25949757 |
| Commensal Bacteria | Harmless oral bacteria are universally carried by healthy adults and can act as reservoirs for genetic adaptation. | PMID 41727074 |
Last updated: April 2026
This article synthesizes findings from peer-reviewed research. It is for educational purposes only and does not constitute medical advice. Consult a healthcare provider before starting any new regimen.



Leave a Reply